Hi Beauties!
Today we're chatting about lash serums and the hormone-like compounds that are found in many of them.
Lash growth products (and all beauty and skincare products) are classified as either a pharmaceutical (drug) or a cosmetic.
The only lash serum in the drug category is the glaucoma treatment turned lash serum, Latisse. All the rest are classified as cosmetics.
DRUGS VS COSMETICS
Products classified as drugs are required to disclose all potential side effects to consumers and must get FDA approval before going to market. They're also required to prove their safety and effectiveness. Of course, a drug can still be considered safe despite potential side-effects, so users have to determine whether the benefits outweigh the potential downfalls.
Cosmetic products (all lash serums but Latisse) on the other hand, are mostly self-policed. The FDA has only banned 11 ingredients since the Federal Food, Drug and Cosmetic Act was passed in 1938. This is compared to the over 1400 banned ingredients in the EU.
The FDA does investigate cosmetics products occasionally when adverse effects are reported, however, they have no authority to recall products unless manufacturers voluntarily agree to. Also, unlike drugs, cosmetic manufacturers are not legally required to report adverse effects to the FDA.
“It’s hard to think of a category that is less regulated [than cosmetics]. ... Even pesticides have more,” - Scott Faber, senior vice president for government affairs at the Environmental Watch Group
PROSTAGLANDINS
Bitmaprost, the active ingredient in Latisse, is known as a prostaglandin. Prostaglandins are chemical compounds with hormone-like effects. Common side effects of these compounds are:
- Darkening of the skin around the eyes
- Eyelid inflammation
- Burning, stinging, and eye pain
- Darkening of the iris
- Thinning and loss of eyelashes
- Blurred vision
- Drooping eyelids.
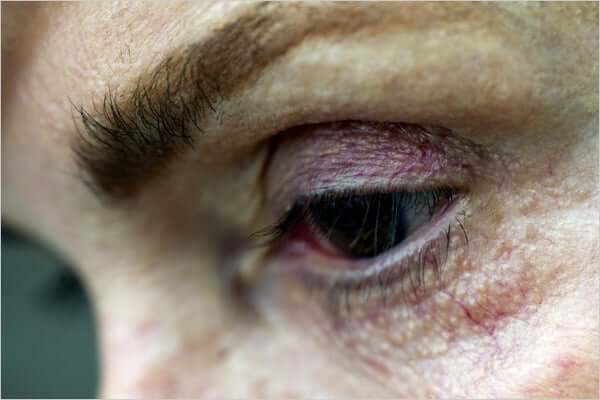
Image: Stephanie Colgan for The New York Times
While consumers are taking a risk with this product, they’re aware of the potential complications when they purchase. The real problem: some lash serums marketed as cosmetics contain a synthetic version of the same prostaglandin analog found in Latisse. These ingredients have the same potential side effects. The difference: these companies are not disclosing the potential side effects to consumers and their formulas are unregulated. No one is making sure these products are safe in the short-term or long-term.
SYNTHETIC PROSTAGLANDINS
One company's serum containing the synthetic prostaglandin, Isopropyl cloprostenate is in the middle of a class-action lawsuit. The company has been accused of "deceptive labeling and unlawful marketing".
The plaintiffs reported symptoms like darkening of the skin around the eyes, eyelid inflammation, burning and stinging, eye pain, darkening of the iris, thinning and loss of eyelashes, blurred vision, and drooping eyelids.
To recognize synthetic prostaglandins, look for these ingredients on product labels:
- Isopropyl cloprostenate
- Isopropanol Phenyl-hydroxyl-pentene Dihydroxy-cyclopentyl-heptenate
- Dechloro Dihydroxy Difluoro Ethylcloprostenolamide
- Trifluoromethyl Dechloro Ethylprostenolamide
Many other lash serum companies also contain synthetic prostaglandins. Some have been recalled and forced to remove these compounds from their formulas. Despite the recalls, however, many brands continue to use synthetic prostaglandins.
POTENTIAL LONG-TERM EFFECTS OF PROSTAGLANDINS
According to Optometrist Dr. Jennifer Lyerly, the inflammation caused by prostaglandin analogs can have lasting negative effects on vision quality and overall eye comfort.
“Every time we blink, the meibomian glands secrete their oil. If that oil is too thick, it's difficult for the eyelids to spread it evenly over the surface of the eye --more like toothpaste than olive oil. It may even get so thick that the oil blocks and backs up in the gland. This will slowly but surely damage the gland permanently. If the meibomian glands atrophy or die off, the body is not able to repair the tissue and the gland becomes permanently nonfunctional. The result is irreversible and often severe dry eye.”
- Dr. Jennifer Lyerly
OTHER TOXIC INGREDIENTS TO AVOID IN YOUR LASH SERUM
Besides prostaglandins, keep an eye out for these toxic chemicals that can affect not just your eyes but your overall health:
PEGS: These ingredients are synthetically created with Ethylene Oxide, a known carcinogen. They can also be contaminated with1,4-Dioxane, another documented carcinogen.
Sodium benzoate + ascorbic acid: Combined, these ingredients can form a carcinogen called benzene.
Potassium Sorbate: classified as a human skin toxicant or allergen by the Cosmetic Ingredient Review Assessments.
Triethanolamine: According to The Derm Review, this ingredient should never be used long term, as it has been proven to have detrimental effects on the skin, the immune system, and has been classified as a respiratory toxicant. These risks are magnified for the sensitive skin around the eyes.
Boric Acid: There’s strong evidence linking boric acid to endocrine disruption.
Sodium Borate: Classified as ‘expected to be toxic or harmful’, sodium borate is restricted for use in cosmetics in Canada and other countries.
OUR LASH & BROW ENHANCING SERUM
Our Lash & Brow Enhancing Serum is 100% natural, non-toxic, hormone-free, and super effective. It's gentle enough to use on your lower lashes too and it will not cause irritation (even if it gets in your eyes!)
For more on how our serum works to encourage longer, thicker lashes (and bolder brows) read our blog.

SWITCHING FROM PROSTAGLANDIN-BASED SERUMS TO A NATURAL SERUM
What most people don't know: when your lashes become accustomed to this external supply of hormones, your lashes may shed rapidly when you stop using the product. Why? Your lash follicles are trying to return to their natural function without the added hormones.
That's why we recommended waiting at least 3 months when switching from a prostaglandin-based serum to our Lash & Brow Enhancing Serum. This will give enough time for your lashes to return to normal.
WHAT YOU CAN DO TO SUPPORT TRANSPARENCY AND SAFETY IN COSMETICS
Hopefully, someday soon, all cosmetics will be completely safe and we won't have to scour ingredients labels for potential toxins. Sadly, that's not yet the case. In order to get there though, two things need to happen:
- Stricter ingredient standards & regulation
- Labeling transparency (all ingredients disclosed + potential side-effects)
Besides voting with your dollars, there are a few ways we can get involved as consumers:
In the US
The Personal Care Products Safety Act is a proposed bill to amend the Federal Food, Drug, and Cosmetic act for the first time in 81 years. It would give the FDA more authority to order recall and police unsafe products.
If passed, the first action would be to ban 5 known carcinogens from cosmetics. Thereafter, the FDA would review at least 5 cosmetic ingredients every year. While this falls short of what's needed (and doesn't come near the regulations in other countries), it is a great start.
To help move this bill forward: contact your local senator with your support for this legislation.
In Canada
Canada has created a Cosmetic Hotlist which bans over 500 ingredients from cosmetic products and restricts the concentration (or requires warnings) for 80+ ingredients. Health Canada, who oversees this list, does have the power to recall products deemed hazardous to consumers.
This list, while an improvement on US regulation, is still not complete. To request that more ingredients be added, contact Health Canada about any products/ingredients you're concerned about - they investigate consumer complaints and evaluate ingredients regularly.
OUR STANCE ON SIDE-EFFECTS & QUESTIONABLE INGREDIENTS
At this point, there's just no reason to accept negative side-effects or toxic ingredients. You can achieve the same (or better) results with safe, pure, non-toxic ingredients.
As a brand, and also as consumers ourselves, we demand better, we will not sacrifice safety for beauty, ever. Everything we create (and buy) must be safe, high-performing, and animal-friendly. #beautywithoutsacrifice
_____________________________
Thanks for tuning into our blog beauties! If you have any questions or would just like to share, send us a message or leave us a comment below <3
-The Plume Team



